Potassium selenide (K2Se) is an inorganic compound formed from selenium and potassium.
 | |
| Names | |
|---|---|
| IUPAC name Potassium selenide | |
| Other names Dipotassium selenide | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.013.817 |
| EC Number |
|
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| K2Se | |
| Molar mass | 157.16 |
| Appearance | clearish wet crystal[1] |
| Density | 2.29 g/cm3[2] |
| Melting point | 800 °C (1,470 °F; 1,070 K)[3] |
| reacts | |
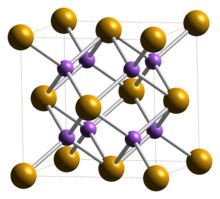
| Structure | |
| cubic: antifluorite | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards | toxic |
| GHS labelling: | |
   | |
| Danger | |
| H301, H331, H373, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+P310, P304+P340, P310, P314, P320, P321, P330, P361, P363, P391, P403+P233, P405, P501 | |
| Related compounds | |
Other anions | Potassium oxide Potassium sulfide Potassium telluride Potassium polonide |
Other cations | Lithium selenide Sodium selenide Rubidium selenide Caesium selenide |
Related compounds | Potassium selenate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Production
It can be produced by the reaction of selenium and potassium. If the two are combined in liquid ammonia, the purity is higher.
Crystal structure
Potassium selenide has a cubic, antifluorite crystal structure.