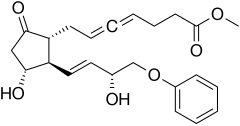
Enprostil is a synthetic prostaglandin designed to resemble dinoprostone. Enprostil was found to be a highly potent inhibitor of gastric HCl secretion.[1] It is an analog of prostaglandin E2 but unlike this prostaglandin, which binds to and activates all four cellular receptors viz., EP1, EP2, EP3, and EP4 receptors, enprostil is a more selective receptor agonist in that it binds to and activates primarily the EP3 receptor.[2] Consequently, enprostil is expected to have a narrower range of actions that may avoid some of the unwanted side-effects and toxicities of prostaglandin E2. A prospective multicenter randomized controlled trial conducted in Japan found combining enprostil with cimetidine was more effective than cimetidine alone in treating gastric ulcer.[3]
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C23H28O6 |
| Molar mass | 400.471 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
See also
References
Further reading
- Toshina K, Hirata I, Maemura K, Sasaki S, Murano M, Nitta M, et al. (December 2000). "Enprostil, a prostaglandin-E(2) analogue, inhibits interleukin-8 production of human colonic epithelial cell lines". Scandinavian Journal of Immunology. 52 (6): 570–575. doi:10.1046/j.1365-3083.2000.00815.x (inactive 2024-04-24). PMID 11119262.
{{cite journal}}: CS1 maint: DOI inactive as of April 2024 (link) - Tari A, Hamada M, Kamiyasu T, Sumii K, Haruma K, Inoue M, et al. (August 1997). "Effect of enprostil on omeprazole-induced hypergastrinemia and inhibition of gastric acid secretion in peptic ulcer patients". Digestive Diseases and Sciences. 42 (8): 1741–1746. doi:10.1023/A:1018825902055. PMID 9286243. S2CID 25069361.
- Ching CK, Lam SK (October 1995). "A comparison of two prostaglandin analogues (enprostil vs misoprostol) in the treatment of acute duodenal ulcer disease". Journal of Gastroenterology. 30 (5): 607–614. doi:10.1007/BF02367786. PMID 8574332. S2CID 6288648.