Elfen gemegol yw magnesiwm a gaiff ei gynrychioli gyda'r symbol Mg a'r rhif atomig 12 yn y tabl cyfnodol. Mae'r elfen yn fetel arian, caled gyda dwysedd isel o 1.738 g/cm³, felly defnyddir y metel fel rhan o nifer o aloïau ysgafn.
| Magnesiwm |
|---|
 | | Magnesiwm mewn cynhwysydd |
|
|---|
 |
|---|
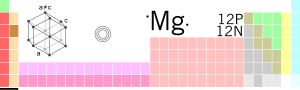
| Symbol | Mg |
|---|
| Rhif | 12 |
|---|
| Dwysedd | 1.738 g/cm³ |
|---|
| Electronegatifedd | 1.31 |
|---|
Magnesiwm |
| Enghraifft o'r canlynol | elfen gemegol, lithophile  |
|---|
| Math | Elfen cyfnod 3, metel daear alcalïaidd, sylweddyn syml  |
|---|
| Deunydd | magnesite, dolomite, brucite, carnallite, talc, Olifin  |
|---|
| Màs | 24.305 ±0.002 uned Dalton  |
|---|
| Fformiwla gemegol | Mg  |
|---|
| Dyddiad darganfod | 1755  |
|---|
| Symbol | Mg  |
|---|
| Rhif atomig | 12  |
|---|
| Trefn yr electronnau | 1s² 2s² 2p⁶ 3s², [Ne] 3s²  |
|---|
| Electronegatifedd | 1  |
|---|
| Cyflwr ocsidiad | 1  |
|---|
| Rhan o | Elfen cyfnod 3, metel daear alcalïaidd  |
|---|
 Ffeiliau perthnasol ar Gomin Wicimedia Ffeiliau perthnasol ar Gomin Wicimedia |


